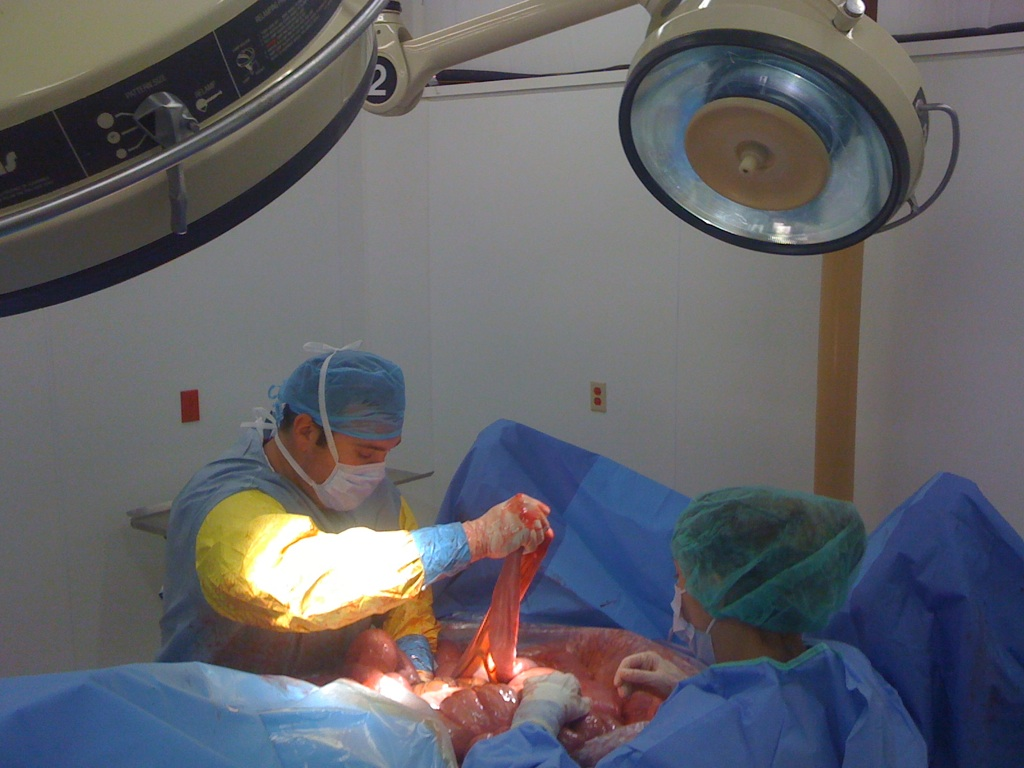
CALEC surgery, a groundbreaking procedure developed at Mass Eye and Ear, is revolutionizing the treatment of severe eye damage through innovative stem cell therapy. This experimental eye surgery involves cultivating autologous limbal epithelial cells, which are crucial for corneal repair, harvested from a healthy eye and transplanted into the damaged eye. In clinical trials, CALEC surgery has demonstrated remarkable efficacy, restoring corneal surfaces for many patients who had previously faced dire visual prospects. With an impressive success rate and a focus on enhancing patients’ quality of life, this approach signifies a beacon of hope for those suffering from various forms of eye damage. As researchers continue to fine-tune this technique, the potential for expanding its applications and accessibility grows, paving the way for new treatments in ocular medicine.
The cultivated autologous limbal epithelial cell (CALEC) technique represents a novel approach to eye treatment, offering new avenues for individuals with debilitating corneal injuries. By harnessing the regenerative power of stem cells, this advanced surgical method not only addresses visual impairments but also focuses on alleviating persistent pain associated with eye damage. This innovative stem cell-based strategy aims to restore the integrity of the cornea, making it a significant development in the realm of ocular repair and rehabilitation. With ongoing trials and studies, practitioners are optimistic that further advancements will lead to wider adoption and success among patients in need of effective eye damage treatment.
Introducing CALEC Surgery for Corneal Repair
The cultivated autologous limbal epithelial cell (CALEC) surgery is pioneering a new era in eye damage treatment. Developed at Mass Eye and Ear, this innovative technique harnesses the healing power of stem cells harvested from a healthy eye to restore the damaged corneal surface in patients suffering from severe eye injuries. Recent clinical trials have demonstrated CALEC’s remarkable effectiveness, with over 90% success in restoring corneal integrity. This opens up new therapeutic avenues for individuals who have faced untreatable corneal damage, primarily due to limbal epithelial cell deficiency.
Ula Jurkunas, an influential figure in this research, emphasizes that CALEC surgery provides significant hope to patients experiencing debilitating corneal injuries which previously left them with limited options. By focusing on stem cell therapy, this approach not only highlights the importance of regenerative medicine but also showcases the intricate processes involved in corneal repair. Through CALEC surgery, patients can regain a clearer vision and a pain-free life, a possibility once thought unreachable for many.
The Role of Stem Cell Therapy in Eye Damage Treatment
Stem cell therapy is revolutionizing the landscape of ocular medicine, especially in the realm of eye damage treatment. The CALEC procedure represents a groundbreaking application of stem cell technology, utilizing limbal epithelial cells to mend corneal surfaces. This method exemplifies the transformative potential of stem cells, which are essential for the regeneration of damaged tissues. In patients with corneal injuries, the depletion of these cells leads to persistent visual impairment and discomfort, placing them in dire need of effective treatments.
The clinical success of stem cell therapies, particularly the CALEC approach, is supported by a range of preclinical studies and rigorous clinical trials. These trials have shown promising outcomes, including a notable restoration of visual acuity and reduction of symptoms related to corneal damage. The ability of stem cell therapy to address conditions previously deemed untreatable marks a significant milestone in eye care, paving the way for further advancements in regenerative techniques aimed at curing ocular diseases.
Mechanism Behind Cultivated Autologous Limbal Epithelial Cells
The mechanism of cultivated autologous limbal epithelial cells (CALEC) entails a meticulous process starting with the extraction of healthy stem cells from a patient’s unaffected eye. This biopsy enables the cultivation of limbal epithelial cells in a controlled environment, where they multiply to form a cellular graft. After a period of two to three weeks, these cells are prepared for transplantation, offering a customized solution to patients suffering from extensive corneal damage. This innovative approach to corneal repair brings hope to those with chronic visual impairment.
During the CALEC procedure, the carefully crafted graft is then surgically implanted into the damaged eye, where it serves to restore the cornea’s protective layer. This transplantation process significantly improves recovery outcomes, as indicated by the trial results showing complete restoration in over 50% of participants within three months. Continuous research into the mechanism of action for CALEC not only enhances our understanding of its efficacy but also reaffirms the potential for stem cell-based therapies to redefine the treatment of various eye injuries.
Limitations and Future of CALEC Surgery
While CALEC surgery offers promising results, certain limitations must be addressed to maximize its potential. One primary restriction is the necessity of having a healthy eye available for biopsy, ruling out patients with bilateral damage. This limitation presents a challenge in expanding the application of CALEC. However, researchers, including Jerome Ritz, are actively exploring allogeneic manufacturing processes using cadaveric donor eyes, which could significantly broaden the reach of this innovative treatment.
As research progresses, future studies are essential for validating CALEC’s effectiveness across diverse patient populations. Increased sample sizes, longitudinal studies, and randomized-control trials will be critical in establishing robust data that could lead to widespread FDA approval. Continued investment in CALEC and similar stem cell therapies could ultimately transform therapeutic practices in eye care, allowing more patients to benefit from advanced regenerative treatments.
Clinical Trial Overview and Results for CALEC Surgery
The recent clinical trials conducted at Mass Eye and Ear were exemplary in demonstrating the efficacy of CALEC surgery. With 14 participants monitored over an 18-month period, results showed that the procedure successfully restored corneal surfaces to an impressive rate of 93% and 92% after 12 and 18 months, respectively. These findings affirm the potential of stem cell therapy not just as a theoretical approach, but as a practical solution for corneal restoration.
Additionally, the trial has highlighted improvements in visual acuity among all participants, showcasing the comprehensive benefits of CALEC surgery beyond just physical repair. While minor adverse events were reported, the overall safety profile reinforces the viability of this treatment, paving the way for future investigations. This trial stands as a significant milestone in the evolution of eye damage treatment and sets the stage for further explorations into stem cell applications in ocular health.
Safety Profile of CALEC Surgery and Follow-Up Care
One of the key components of evaluating any new surgical intervention is its safety profile. The CALEC surgery demonstrated a high level of safety during the clinical trial, with no serious adverse events reported among the study participants. The only noted complication was a bacterial infection, considered minor in nature and previously associated with chronic contact lens use. Understanding the safety outcomes is crucial for both practitioners and patients as they consider this novel procedure.
Follow-up care for patients undergoing CALEC surgery remains integral to ensuring long-term success and monitoring recovery. Regular assessments of the corneal surface post-surgery allow for prompt recognition of any complications and the opportunity for timely interventions. As the protocol for CALEC surgery evolves, establishing robust follow-up care protocols will be critical in cementing its role in the treatment landscape for corneal damage.
Impacts of CALEC on Visual Acuity and Quality of Life
Beyond the technical success of restoring the cornea, CALEC surgery has profound implications for the visual acuity and overall quality of life of patients. The results of the clinical trials indicated substantial improvements in vision, offering hope to those who endured the persistent challenges of corneal damage. Enhanced visual acuity translates not only to a more fulfilling daily life but also to increased independence and self-esteem for patients previously hampered by their eye injuries.
By effectively addressing the root causes of corneal injury through stem cell therapy, CALEC offers a lifeline to individuals who had limited options for regaining their sight. As outcomes continue to improve with ongoing research, the holistic impact of successful corneal restoration can lead to enriched lives, fostering a community of patients empowered by their ability to see clearly once again.
Regulatory Journey Ahead for CALEC Surgery
Although the promising results from the CALEC clinical trials are encouraging, the pathway toward FDA approval is a significant journey. With the necessity of additional studies involving larger patient cohorts and extended follow-ups, the research team at Mass Eye and Ear is committed to advancing this innovative therapy. Their focus on generating comprehensive data will be pivotal in navigating the regulatory landscape and ensuring efficient transition from experimental to widely accepted treatment.
The future of CALEC surgery not only hinges on scientific validation but also on continued advocacy for support from funding bodies and health organizations. As the clinical landscape continues to evolve with new findings, the efforts to integrate CALEC into standard treatment options will be a concerted one, necessitating collaboration among researchers, clinicians, and regulatory bodies.
Prospects for Allogeneic Limbal Stem Cell Approaches
The vision for expanding the use of CALEC surgery hinges on the development of allogeneic limbal stem cell therapies. By sourcing stem cells from cadaveric donor eyes, researchers hope to create a more accessible option for patients suffering from bilateral corneal injuries. This shift could revolutionize the way corneal repairs are conducted, making CALEC surgery available to a broader range of patients.
The potential for allogeneic approaches highlights an exciting frontier in regenerative medicine. With ongoing advancements in stem cell research and tissue engineering, the application of CALEC could serve as a model for other areas of regenerative medicine. This pioneering effort underscores the pressing need for continued investment in innovative eye damage treatments to ensure that patients have access to effective therapies that can restore their vision and quality of life.
Frequently Asked Questions
What is CALEC surgery and how does it relate to stem cell therapy?
CALEC surgery, or cultivated autologous limbal epithelial cell surgery, involves transplanting stem cells from a healthy eye to repair corneal damage. This experimental eye surgery uses stem cell therapy to regenerate limbal epithelial cells, providing new hope for individuals with severe corneal injuries that were once considered untreatable.
How effective is CALEC surgery in treating corneal damage?
CALEC surgery has shown a remarkable effectiveness rate, with over 90% of participants in a clinical trial experiencing successful restoration of the cornea’s surface. The procedure is particularly effective in providing relief and improving vision for patients suffering from eye damage due to injuries like chemical burns or infections.
Who can benefit from CALEC surgery?
Patients with limbal stem cell deficiency due to corneal injuries may benefit from CALEC surgery. This innovative treatment is designed for those who have one healthy eye from which stem cells can be harvested. Future developments hope to allow treatment for patients with bilateral eye damage through allogeneic procedures.
What are the safety considerations associated with CALEC surgery?
During clinical trials of CALEC surgery, no serious adverse events were reported in donor or recipient eyes. The treatment displayed a high safety profile, although there was a minor incidence of a bacterial infection related to post-operative care. Overall, the risks are considered low compared to the potential benefits of restoring vision.
Is CALEC surgery currently available for patients?
As of now, CALEC surgery remains an experimental treatment and is not widely available at Mass Eye and Ear or other U.S. hospitals. Ongoing research aims to expand access to this procedure, with hopes for FDA approval following further clinical trials.
What does the future hold for CALEC surgery and stem cell therapy in corneal repair?
The future of CALEC surgery looks promising as researchers plan larger-scale trials to enhance the treatment’s applicability. The ongoing studies seek to establish protocols that would enable FDA approval, potentially expanding this groundbreaking stem cell therapy to a larger pool of patients facing corneal damage.
How does CALEC surgery compare with traditional corneal transplants?
Unlike traditional corneal transplants, which rely on donor corneas, CALEC surgery uses a patient’s own stem cells, minimizing rejection risks. This innovation offers a groundbreaking solution for patients who might not be candidates for conventional transplants due to existing damage or defects.
| Key Points |
|---|
| First CALEC surgery performed by Ula Jurkunas at Mass Eye and Ear, marked significant advances in eye damage treatment. |
| Stem cell therapy using cultivated autologous limbal epithelial cells (CALEC) restores cornea surfaces in patients with blinding injuries. |
| Clinical trial involving 14 patients showed over 90% effectiveness in restoring corneal surface after 18 months. |
| Procedure involves harvesting stem cells from a healthy eye, growing them, and transplanting to the damaged eye. |
| Significant improvement in visual acuity noted in patients, with no major adverse events reported. |
| The clinical trial was the first human study funded by the National Eye Institute focusing on stem cell therapy for eye treatment. |
| Future studies aim to include larger patient groups and other centers to validate findings and facilitate FDA approval. |
Summary
CALEC surgery represents a groundbreaking advancement in eye health, offering new hope to patients with previously untreatable corneal injuries. The promising results from the clinical trial indicate that this innovative stem cell therapy not only effectively repairs corneal surfaces but also significantly improves overall vision for patients. As further research unfolds, the goal remains to make CALEC surgery widely accessible, with the potential to transform the standard of care for individuals suffering from severe eye damage.